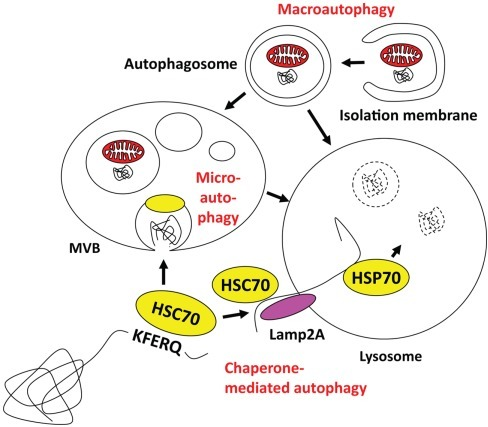 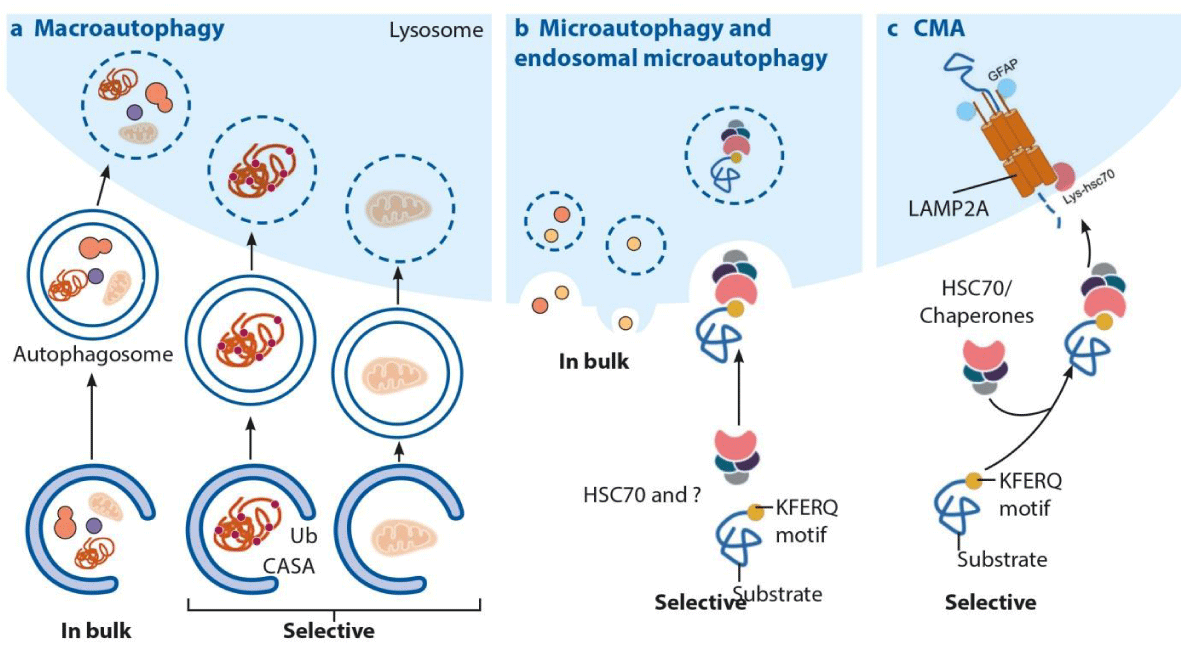
PHHs were cultured in a six-well tissue culture plate using growth media supplied by the XenoTech LLC plus 10% human serum (Invitrogen, Brown Deer, WI). We report a novel mechanism through which stress response triggers oncogenic Nrf2 signaling that promotes autophagy switching to favor cell survival. Silencing Nrf2 and LAMP2A reduced cell viability, suggesting that the stress response activates CMA as a compensatory mechanism of cell survival. This impaired autophagosome-endosome fusion due to beclin1 degradation inhibited endocytosis and degradation of epidermal growth factor receptor. CMA selectively targeted beclin1 degradation, leading to accumulation of the autophagy flux protein p62 due to impaired autophagosome-endosome fusion. Protein kinase R–like endoplasmic reticulum kinase–dependent activation of nuclear factor erythroid 2–related factor (Nrf2) led to increased transcription of the cytoprotective genes: heat shock cognate 70 kDa protein and lysosome-associated membrane protein 2A (LAMP2A) and precipitated the induction of CMA. Protein kinase R–like endoplasmic reticulum kinase expression increased to high levels in hepatitis C virus culture. This study examined how the stress response executes oncogenic cell programming through autophagy switching using hepatitis C virus cell culture. We found that HCC grown in the highly stressed cirrhotic microenvironment undergoes autophagy switching from a protective state characterized by high macroautophagy with low chaperone-mediated autophagy (CMA) to an HCC-promoting state characterized by low macroautophagy with high CMA. The mechanisms that contribute to HCC development in the cirrhotic microenvironment are unknown. Liver cirrhosis is an independent risk factor for hepatocellular carcinoma (HCC).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
